A peptide bond is a CO-NH bond formed between two molecules, occuring when the carboxyl group of one molecule reacts with the amino group of the other molecule
Peptide bond definition
This is a dehydration synthesis reaction (also known as a condensation reaction), and usually occurs between amino acids. The resulting CO-NH bond is called a peptide bond, and the resulting molecule is an amide. The four-atom functional group -C(=O)NH- is called an amide group or (in the context of proteins) a peptide group. Polypeptides and proteins are chains of amino acids held together by peptide bonds, as is the backbone of PNA.
The peptide group (CO–NH) has a rigid planar structure. This structure is due to interactions between electrons of the double bond of the carbonyl group and those of the C–N bond such that the latter acquires partial (about 40%) double-bond properties.

Since single bonds between two atoms are longer than double bonds between the same two atoms, the lengths of the C–N and C=O bonds in the peptide group differ from those observed for these bonds in other contexts where resonance does not occur. Thus the partial double bond character of C–N in the peptide group means that this bond is shorter than would be predicted for a C–N single bond, whilst the C=O bond, having a partial single bond character due to resonance, is longer than would be predicted for a C=O double bond.
Generally speaking, peptide bonds are in the trans conformation. However, cis forms can occur in peptide bonds that precede a proline residue. In such cases, the cis form is more stable than usual since the proline side-chain offers less of a hindrance. Nonetheless, cis peptide bonds occur only in approximately 10% of instances of peptide bonds preceding proline residues.
Peptide bond formation
Peptide bond example
The amino acids in the ribosome are attached to their respective tRNAs by an ester bond (R – O – R) between the carboxyl terminus and the amino acceptor stem (left). During formation of a peptide bond, the ester bond in the (P)eptidyl site is cleaved, and Peptidyl Transferase catalyzes a condensation reaction between its carboxyl terminus and the amino terminus of the amino acid in the (A)mino site. This transfers the P-site amino acid to the A-site amino acid, and the original amino terminus remains unmodified. The polypeptide thus “grows” from the amino terminus to the carboxyl terminus.
Find your dream job in the space industry. Check our Space Job Board »
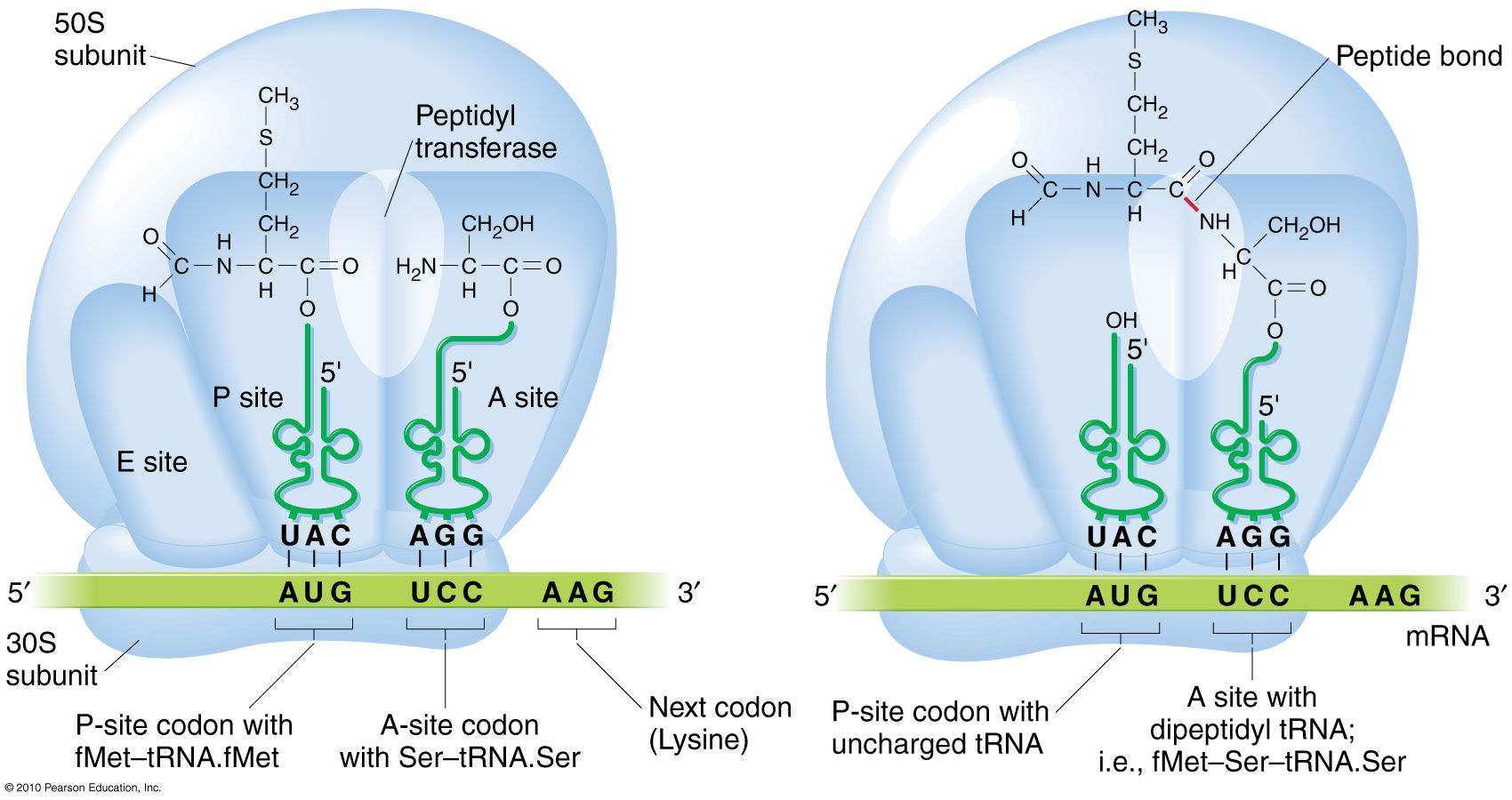
In vitro formation of a peptide bond is described as a dehydration reaction that splits out of an H20. However, the in vivo reaction is a condensation reaction. Because the C-terminus of the amino acid in the P site is joined to the 3′ terminus of the tRNA by an ester bond, it participates in peptide bond formation as a carbonyl radical (-C=O) without an –OH radical. When the C-terminus joins with the NH2 terminus of the amino acid in the A site, the end result is the shift of a proton (-H) from the amino terminus to the uncharged tRNA molecule. This balances the reaction, and forms a peptide bond without release of an H20 molecule.





