Magnesium dimer (Mg2) is a fragile molecule consisting of two weakly interacting atoms held together by the laws of quantum mechanics. It has recently emerged as a potential probe for understanding fundamental phenomena at the intersection of chemistry and ultracold physics, but its use has been thwarted by a half-century-old enigma—five high-lying vibrational states that hold the key to understanding how the magnesium atoms interact but have eluded detection for 50 years.
The lowest fourteen Mg2 vibrational states were discovered in the 1970s, but both early and recent experiments should have observed a total of nineteen states. Like a quantum cold case, experimental efforts to find the last five failed, and Mg2 was almost forgotten. Until now.
Piotr Piecuch, Michigan State University Distinguished Professor and MSU Foundation Professor of chemistry, along with College of Natural Science Department of Chemistry graduate students Stephen H. Yuwono and Ilias Magoulas, developed new, computationally derived evidence that not only made a quantum leap in first-principles quantum chemistry, but finally solved the 50-year-old Mg2 mystery.
Their findings were recently published in the journal Science Advances.
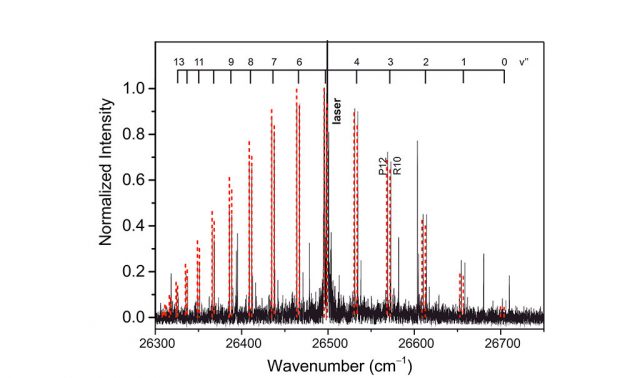
“Our thorough investigation of the magnesium dimer unambiguously confirms the existence of 19 vibrational levels,” said Piecuch, whose research group has been active in quantum chemistry and physics for more than 20 years. “By accurately computing the ground- and excited-state potential energy curves, the transition dipole moment function between them and the rovibrational states, we not only reproduced the latest laser-induced fluorescence (LIF) spectra, but we also provided guidance for the future experimental detection of the previously unresolved levels.”
Find your dream job in the space industry. Check our Space Job Board »
So why were Piecuch and his team able to succeed where others had failed for so many years?
The persistence of Yuwono and Magoulas certainly revived interest in the Mg2 case, but the answer lies in the team’s brilliant demonstration of the predictive power of modern electronic structure methodologies, which came to the rescue when experiments encountered unsurmountable difficulties.
“The presence of collisional lines originating from one molecule hitting another and the background noise muddied the experimentally observed LIF spectra,” Piecuch explained. “To make matters worse, the elusive high-lying vibrational states of Mg2 that baffled scientists for decades dissipate into thin air when the molecule starts rotating.”
Instead of running costly experiments, Piecuch and his team developed efficient computational strategies that simulated those experiments, and they did it better than anyone had before.
Like the quantized vibrational states of Mg2, in-between approximations were not acceptable. They solved the electronic and nuclear Schrödinger equations, tenets of quantum physics that describe molecular motions, with almost complete accuracy.
“The majority of calculations in our field do not require the high accuracy levels we had to reach in our study and often resort to less expensive computational models, but we provided compelling evidence that this would not work here,” Piecuch said. “We had to consider every conceivable physical effect and understand the consequences of neglecting even the tiniest details when solving the quantum mechanical equations.”
Their calculations reproduced the experimentally derived vibrational and rotational motions of Mg2 and the observed LIF spectra with remarkable precision—on the order of 1 cm-1, to be exact. This provided the researchers with confidence that their predictions regarding the magnesium dimer, including the existence of the elusive high-lying vibrational states, were firm.
Yuwono and Magoulas were clearly excited about the groundbreaking project, but emphasized they had initial doubts whether the team would be successful.
“In the beginning, we were not even sure if we could pull this investigation off, especially considering the number of electrons in the magnesium dimer and the extreme accuracies required by our state-of-the-art computations,” said Magoulas, who has worked in Piecuch’s research group for more than four years and teaches senior level quantum chemistry courses at MSU.
“The computational resources we had to throw at the project and the amount of data we had to process were immense—much larger than all of my previous computations combined,” added Yuwono, who also teaches physical chemistry courses at MSU and has worked in Piecuch’s research group since 2017.
The case of the high-lying vibrational states of Mg2 that evaded scientists for half a century is finally closed, but the details of the computations that cracked it are completely open and accessible on the Science Advances website. Yuwono, Magoulas, and Piecuch hope that their computations will inspire new experimental studies.
“Quantum mechanics is a beautiful mathematical theory with a potential of explaining the intimate details of molecular and other microscopic phenomena,” Piecuch said. “We used the Mg2 mystery as an opportunity to demonstrate that the predictive power of modern computational methodologies based on first-principles quantum mechanics is no longer limited to small, few-electron species.”
Provided by: Michigan State University
More information: Stephen H. Yuwono et al. Quantum computation solves a half-century-old enigma: Elusive vibrational states of magnesium dimer found. Science Advances (2020). DOI: 10.1126/sciadv.aay4058
Image: This graph shows the team’s highly accurate ab initio calculations in red, dotted lines relative to the experimental LIF spectrum of Mg2, marked in black.
Credit: Piecuch Lab