University of Minnesota Twin Cities researchers in the Department of Chemistry have created a new polymer to deliver DNA and RNA-based therapies for diseases. For the first time in the industry, the researchers were able to see exactly how polymers interact with human cells when delivering medicines into the body. This discovery opens the door for more widespread use of polymers in applications like gene therapy and vaccine development.
The research is published in the Proceedings of the National Academy of Sciences (PNAS), a peer-reviewed multidisciplinary scientific journal.
Gene therapy involves altering the genes inside the body’s cells to treat or cure diseases. It requires a carrier that “packages” the DNA to deliver it into the cell—oftentimes, a virus is used as a carrier. Packaging of nucleic acids is also used in vaccines, such as the recently developed messenger RNA (mRNA) COVID-19 vaccine, which is enclosed in a lipid.
The research team is led by chemistry professor Theresa Reineke and associate professor Renee Frontiera. Reineke’s lab synthesizes polymers, which are long-chain molecules that make up plastics, to use for packaging the nucleic acids instead.
“It’s kind of like ordering something from Amazon, and it’s shipped in a box,” Reineke explained. “Things get broken if they’re not delivered in a package. That’s basically what we’re doing here but on a nano-level. We’re taking these really sensitive RNA and DNA cargo that are susceptible to enzymatic degradation, that won’t get to their target unless you have something to protect them.”
Find your dream job in the space industry. Check our Space Job Board »
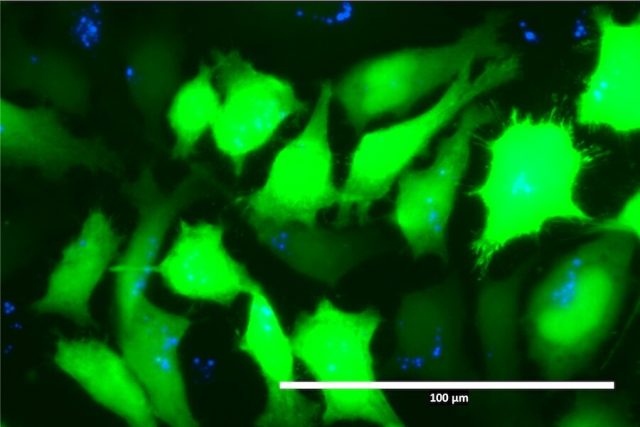
The researchers designed the copolymer using quinine, a naturally occurring substance used in tonic water, and 2-hydroxyethyl acrylate (HEA), which makes the material soluble and is used in a variety of personal care and medical materials. Because quinine is fluorescent, the research team was able to track the DNA package throughout the body and into the cells using Raman spectroscopy, a chemical imaging technique.
“We’ve discovered a new packaging tool with this natural product that’s important for all of these high-flying, important fields like gene therapy and vaccines,” said Reineke, who is also a Distinguished McKnight University Professor. “And, it works in a variety of cell-types. On top of that, it’s got all of these cool features—it’s fluorescent, we can track it, it’s Raman active, and that allowed us to understand a lot of fundamentals about these packaging systems that were impossible to probe before we incorporated this natural product.”
Polymer-based drug delivery is significantly cheaper than using viruses, especially for gene therapy, which can cost up to $2 million for a single injection. However, the main barrier preventing widespread polymer use was that scientists didn’t know a lot about how the polymer package actually interacts with cells in the body.
This research helps clear up that uncertainty. Frontiera’s lab specializes in chemical imaging. Using Raman spectroscopy, they discovered that a cell’s own proteins play a key role in unpacking the nucleic acid cargo once the polymer carrier enters the cell.
“It’s very satisfying to know how this is actually happening, what the process of delivery is, and to actually see that in real-time,” Frontiera said. “A key point is that these polymers also work very well. For all the beneficial attributes, they’re also incredibly effective at getting the payload into cells, and we were able to tell why, which doesn’t always happen in this field.”
Provided by: University of Minnesota
More information: Craig Van Bruggen et al. Quinine copolymer reporters promote efficient intracellular DNA delivery and illuminate a protein-induced unpackaging mechanism. Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.2016860117
Image: The above image shows cells illuminated by quinine (fluorescent blue) and green fluorescent protein. This allows the University of Minnesota researchers to—for the first time—observe how polymers interact with human cells when delivering medicines. The discovery could be used in advancing DNA and RNA-based treatments such as gene therapy and vaccines.
Credit: Reineke Group and Frontiera Group, University of Minnesota