The nervous system is loaded with encoded information: thoughts, emotions, motor control. This system in our bodies is an enigma, and the more we can do to understand it, the more we can do to improve human life. Brain-machine interfaces provide one way to connect with this puzzling organ system, including the brain. But because electronic devices are stiff, planar, and rigid, they cause injury to the brain’s soft tissue.
Until now, it’s been extremely challenging to develop a material and fabrication method that is flexible enough to meld with the brain, but adhesive enough to stay in one place. However, Carnegie Mellon University Associate Professor of Materials Science and Engineering and Biomedical Engineering Chris Bettinger and his group have created a hydrogel material and fabrication process for electrodes that stick to the brain, matching its soft, squishy makeup.
“Imagine you have a bowl of Jell-O, and you insert a rigid plastic fork into the bowl and move it around,” says Bettinger. “It’s going to damage the Jell-O, producing defects and irreversible structural changes. That situation is analogous to inserting a rigid electronic probe into soft tissue such as someone’s brain. It’s a combination of what we call micro-motion and mechanics, which work together to not only damage the brain, but also compromise the function of the implanted sensor.”
The rigid electrode detects when neurons are firing and records the voltages associated with those firing neurons. But over time, the body interprets this material as an injury and a foreign body that needs to be attacked, degraded, isolated, and removed. Inflammatory cells then surround the probe, disrupting the signal strength of the neurons in that area.
In the past twenty years, silicon-based electronics have progressed from rigid and planar in form to curved, flexible, and stretchable. The rigidity of these electronics has evolved from being stiff like wood, to thin and flexible like paper, to stretchy and pliable like rubber bands. Now, Bettinger’s team is taking it one step further, making them not only flexible and stretchable, but also extremely soft and adhesive.
Find your dream job in the space industry. Check our Space Job Board »
“If we could fabricate electronic devices that have mechanical properties closer to ‘Jell-O’ rather than wood or plastic, then we can surreptitiously interface neural probes with the brain in a more benign manner,” says Bettinger.
The challenge is that the processes used to make sophisticated electronics require high temperatures (400 C or higher), a vacuum, and exotic solvents, buffers, acids, and bases to etch materials and patterns. None of these are compatible with soft hydrogel materials.
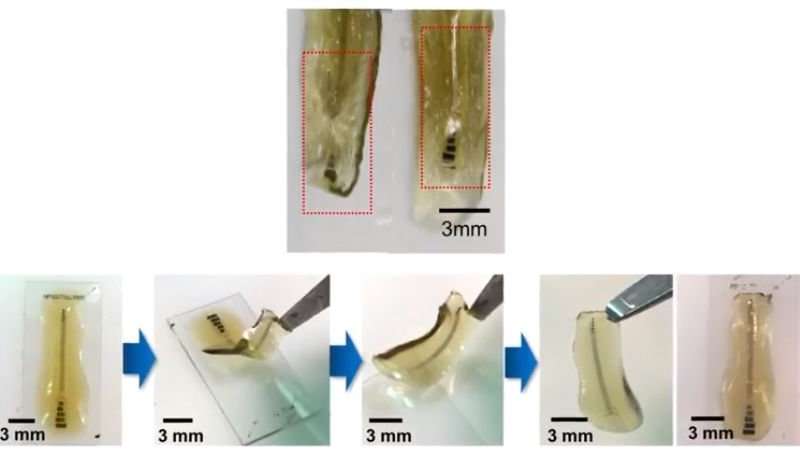
To combat these fundamental issues, Bettinger and his team created a new way to fabricate the electronics—decoupling the fabrication processes of the electronic part and the soft substrate it’s embedded in. First, they construct the electronic part on a substrate that is compatible with high temperatures, extreme solvents, and a vacuum, and create the hydrogel substrate separately. Then, they remove the electronic piece from its original substrate and adhere it to the hydrogel substrate. The final device contains a thin layer of electronics on a soft, flexible, and sticky substrate that has mechanical properties similar to those of the nervous system.
Another challenge was creating a material that was still adhesive in fluid. If the material can’t adhere when wet, it would be like trying to keep a Band-Aid on while in the pool. For the electrode to work it needs to stick in one place for a long period of time. The researchers studied the properties of animals like the blue mussel, which sticks to rocks underwater. They applied those same chemical principles when creating the hydrogel substrate.
“Instead of having to take a brain or a spinal cord and then stick something into it and then injure it,” says Bettinger, “we can laminate it on top and avoid injury to the tissue.”
The fact that the nodes do not injure the tissue and do not move around means that they are able to record a stronger and more accurate signal from the firing neurons. The probes could now be used not only to record signals, but also to stimulate therapies.
For example, the electrode array in the probe could block the signal that induces inflammation in people with rheumatoid arthritis. Instead of using painkillers like opiates, an electronic-based therapy that stimulates appropriate regions of the spinal cord could be more targeted and effective, while avoiding the risk of addiction when compared to pharmaceutical-based interventions. The electrodes can also be used for long-term recording applications, such as testing how a new drug could affect the heart. A sticky, soft electrode that can bend and flex can ensconce the heart, record its contractions, and indicate which drug might be most effective.
“We’re trying to improve the temporal bandwidth of these probes, by preserving the longevity of the material. Then we can acquire more information and maintain a suitable signal-to-noise ratio,” said Bettinger. “Researchers in multiple disciplines are trying to improve the way that electronic devices can interface with the nervous system. We feel that we are contributing to this broader effort by expanding the materials toolbox to improve device performance.”
Bettinger and his group are collaborating with researchers in electrical and computer engineering at Carnegie Mellon and with researchers at the University of Pittsburgh. Their findings have been published inAdvanced Functional Materials.
Provided by:
Carnegie Mellon University Materials Science and Engineering
More information:
Wei-Chen Huang et al. Ultracompliant Hydrogel-Based Neural Interfaces Fabricated by Aqueous-Phase Microtransfer Printing. Advanced Functional Materials (2018). DOI: 10.1002/adfm.201801059
Image:
Credit: College of Engineering, Carnegie Mellon University