The discovery last year of the first nickel oxide material that shows clear signs of superconductivity set off a race by scientists around the world to find out more. The crystal structure of the material is similar to copper oxides, or cuprates, which hold the world record for conducting electricity with no loss at relatively high temperatures and normal pressures. But do its electrons behave in the same way?
The answers could help advance the synthesis of new unconventional superconductors and their use for power transmission, transportation and other applications, and also shed light on how the cuprates operate—which is still a mystery after more than 30 years of research.
In a paper published today in Nature Materials, a team led by scientists at the Department of Energy’s SLAC National Accelerator Laboratory and Stanford University report the first detailed investigation of the electronic structure of superconducting nickel oxides, or nickelates. The scientists used two techniques, resonant inelastic X-ray scattering (RIXS) and X-ray absorption spectroscopy (XAS), to get the first complete picture of the nickelates’ electronic structure—basically the arrangement and behavior of their electrons, which determine a material’s properties.
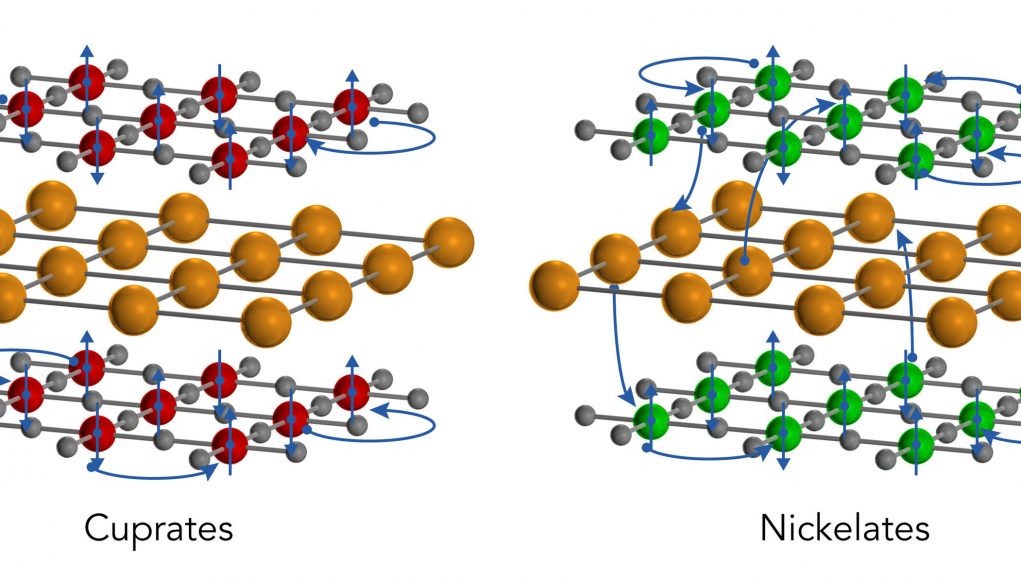
Both cuprates and nickelates come in thin, two-dimensional sheets that are layered with other elements, such as rare-earth ions. These thin sheets become superconducting when they’re cooled below a certain temperature and the density of their free-flowing electrons is adjusted in a process known as “doping.”
Cuprates are insulators in their predoped “ground” states, meaning that their electrons are not mobile. After doping the electrons can move freely but they are mostly confined to the cuprate layers, rarely traveling through the intervening rare-earth layers to reach their cuprate neighbors.
Find your dream job in the space industry. Check our Space Job Board »
But in the nickelates, the team discovered, this is not the case. The undoped compound is a metal with freely flowing electrons. Furthermore, the intervening layers actually contribute electrons to the nickelate sheets, creating a three-dimensional metallic state that is quite different from what’s seen in the cuprates.
This is an entirely new type of ground state for transition metal oxides such as cuprates and nickelates, the researchers said. It opens new directions for experiments and theoretical studies of how superconductivity arises and how it can be optimized in this system and possibly in other compounds.
Provided by: SLAC National Accelerator Laboratory
More information: M. Hepting et al. Electronic structure of the parent compound of superconducting infinite-layer nickelates. Nature Materials (2020). DOI: 10.1038/s41563-019-0585-z
Image: A SLAC/Stanford study found that a recently discovered family of nickelate superconductors differs in surprising ways from a related family, the cuprates. Both come in 2D oxide planes (red, green, and grey spheres representing copper, nickel and oxygen ions, respectively) separated by layers of a rare earth material (gold spheres). Cuprates are inherently insulators, and even when they’re doped to add free-flowing electrons (blue spheres), as shown here, their electrons rarely leave to interact with other layers of material. But these nickelates are inherently metals. Even in the non-doped state depicted here, their electrons mix with electrons from the rare-earth layers in a way that creates a 3D metallic state.
Credit: Greg Stewart/SLAC National Accelerator Laboratory